Your risk for heart disease in a single number
Debate continues on over how much genes influence risk of heart attack
Marcus Banks • March 5, 2019
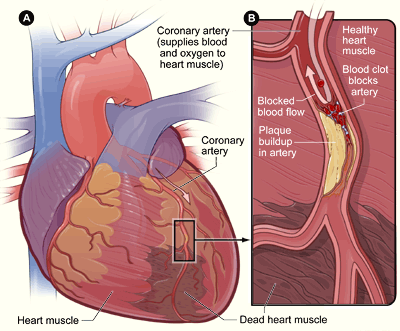
Plaque build-up in arteries can cause a heart attack. While regular exercise and a healthy diet are crucial to reducing chances of heart attack, some of the risk may lie in our genes. [Source: US National Heart, Lung and Blood Institute | Public Domain]
Why do young and healthy people with no apparent risk of heart trouble sometimes have heart attacks? Dr. Sekar Kathiresan, a cardiologist in Boston, thinks the answer lies hidden in their genes. He is working to capture this risk in a single number.
Kathiresan’s research team, based at Massachusetts General Hospital and the Broad Institute in Cambridge, Mass., has developed a “polygenic risk score” to detect whether a person has an increased risk for heart attack based on variations in their genes. They reported their findings last fall in the journal Nature Genetics.
Polygenic risk scores will never be bull’s-eye predictors of who will have a heart attack and who will not, Kathiresan says. A person’s habits and choices, as well as what they are exposed to in their environment, will always play major roles in determining heart attack risk. “DNA is not destiny,” he says.
Those limits acknowledged, Kathiresan argues that risk scores can still save and improve lives. The sooner people know about their genetic risks, he says, the sooner they can take action to reduce their chances of becoming one of the 735,000 Americans who suffer a heart attack every year.
But some scientists argue that Kathiresan is oversimplifying a complex problem. One critic is Cecile Janssens, an epidemiologist at Emory University in Atlanta, Georgia. Janssens and Kathiresan have engaged in heated Twitter exchanges about this topic, and in 2018 discussed the value of polygenic risk scores at a debate organized by the Centers for Disease Control and Prevention. Janssens argues that polygenic risk scores are mostly an elaborate means of “adding up many small effects” that may not predict anything meaningful.
In the Nature Genetics report, Kathiresan and colleagues examined more than six million points on the genomes of 289,000 participants in the UK Biobank project. They found that individuals with the most genetic variations associated with coronary heart disease were three times as likely to develop the disease as people with fewer of those variations. Coronary artery disease is a leading cause of heart attack.
Boiling down a health risk into one easily understood number — for example, three times as likely to develop coronary artery disease — is the essence of a polygenic risk score. Eight percent of UK Biobank participants reached this threshold of risk in Kathiresan’s study. This means that nearly one in 10 people in his sample had an elevated risk of heart disease based on their genetic profile.
This finding contrasts sharply with the most commonly available genetic test now used to identify heart disease risk, which finds elevated risk in less than one percent of people.
The type of genetic variation Kathiresan studies is called a single nucleotide polymorphism. SNPs occur when there is a change in a single molecule on a strand of DNA, which is composed of four molecules indicated by the letters C, A, G and T. One SNP, for example, would be C-A-G-A changing to C-A-G-T. While most SNPs have little or no impact on human health, some are associated with medical conditions like coronary artery disease.
SNPs associated with coronary heart disease are well understood, so they are the basis for Kathseran’s polygenic risk scores. Web-based tools that seek to present people with their risk for coronary artery disease are in the early stages of development. For example, the Scripps Research Institute in California is building a predictor called MyGeneRank that will use data from 23andMe to produce an individualized risk score. Kathiresan’s group is building a similar product. Both efforts are in early prototyping stages, and are not meant to be deployed widely in hospitals and clinics (yet).
Right now the focus is in on heart disease. But in the future, polygenic risk scores could also be used to help clinicians reduce unnecessary screening for other diseases by ensuring that only the people most at risk are tested.
Breast cancer is one possible example, says Lawrence Brody, who heads the Genetics and Environment Interaction Section at the National Human Genome Research Institute. The current consensus is that all women over the age of 50 should receive regular mammograms. This is true even though mammograms are not always accurate and women vary in their chances of developing breast cancer.
While this one-size-fits-all approach is all doctors have to work with now, robust polygenic risk scores could eventually determine which women should be frequently screened and which should not. We are a long way from this point, Brody cautions, and women should follow the current screening guidelines.
No matter how polygenic risk scores evolve, they need to become more reflective of the entire world’s population, says Alicia Martin at the Broad Institute. To date, the large majority of genetic sequencing has occurred in people of European descent, she explains, which means that any analysis based on these sequences are not that applicable to most of the world.
Researchers need to conduct “much larger studies, especially in non-European populations,” Martin says. Once this data exists, she continues, it needs to be just as accessible as data about European groups.
A recent study by Kathiresan and colleagues shows that polygenic risk scores can predict heart attack risk across racial groups — although as of now they are most accurate for white people. This supports Martin’s arguments for much larger studies in other demographic groups.
Kathiresan readily recognizes the diversity challenge, calling it out quite regularly in his papers. But this does not dampen his overall enthusiasm about polygenic risk scores. In the near future, he predicts, doctors will prepare a medical “report card” based on someone’s genes. “When I talk to cardiologists about this, they get super excited,” he says.