The mRNA revolution is just getting started
In the wake of COVID successes, cancer vaccines and other innovative treatments are on the way
Deborah Balthazar • March 7, 2022
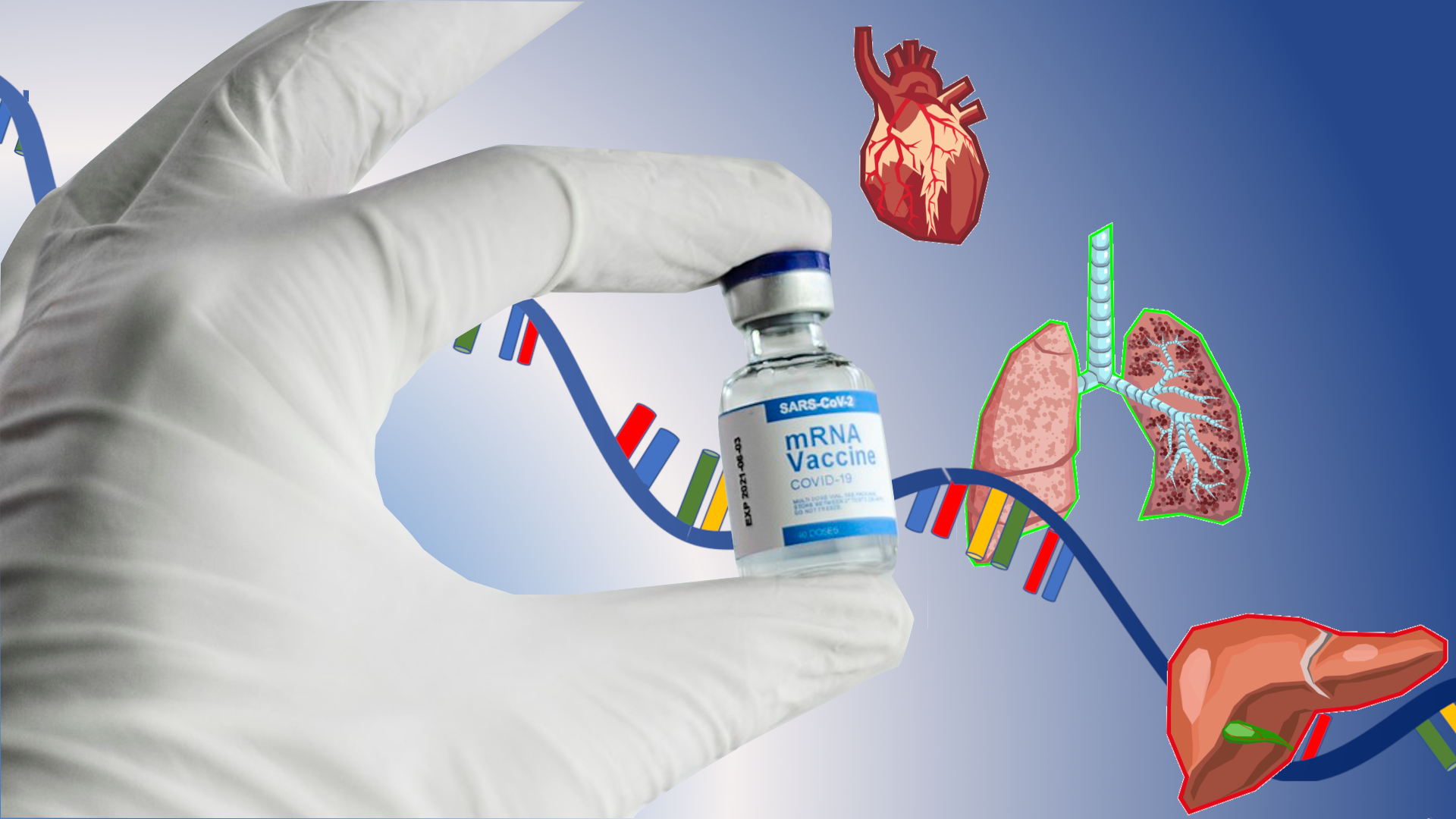
Most people know about mRNA from COVID-19 vaccines, but experts say the technology may soon be used to treat or even prevent a seemingly limitless range of illnesses. [Credit: Deborah Balthazar | vial and glove: Spencer B Davis (CC BY 4.0) | Organ images: mothsart (CC0 1.0)]
Imagine a vaccine for any cancer, or a wound that heals without leaving a scar. You may not need to imagine much longer: A bounty of treatments using technology similar to two of the most successful COVID-19 vaccines is coming soon, scientists and biotechnology experts predict.
Hoping to match the success of the Moderna and Pfizer/BioNTech vaccines, other companies are now taking shots at creating mRNA therapies for rare cancers, autoimmune disorders and other diseases. But the therapies will only make a difference if companies can figure out how to scale up these personalized therapies to as many patients as possible.
“The possibilities are nearly limitless,” says cardiologist Dr. John Cooke, who in March 2021 co-authored a paper with the slightly hyperbolic title, “The Limitless Future of RNA Therapeutics.” These new therapies will be relatively inexpensive to develop and manufacture and will target diseases that until now have been “undruggable,” predicts Cooke, also the medical director for the RNA Therapeutics Program at Houston Methodist Research Institute in Texas.
The sunny optimism from Cooke and others is rooted in the malleability of RNA. Like DNA, RNA is a nucleic acid that carries genetic instructions but is cut in half. The single-stranded nature of RNA makes it easier to manipulate. The new therapies all target a particular type of RNA — messenger RNA, or mRNA — which carries the instructions for protein production in our cells.
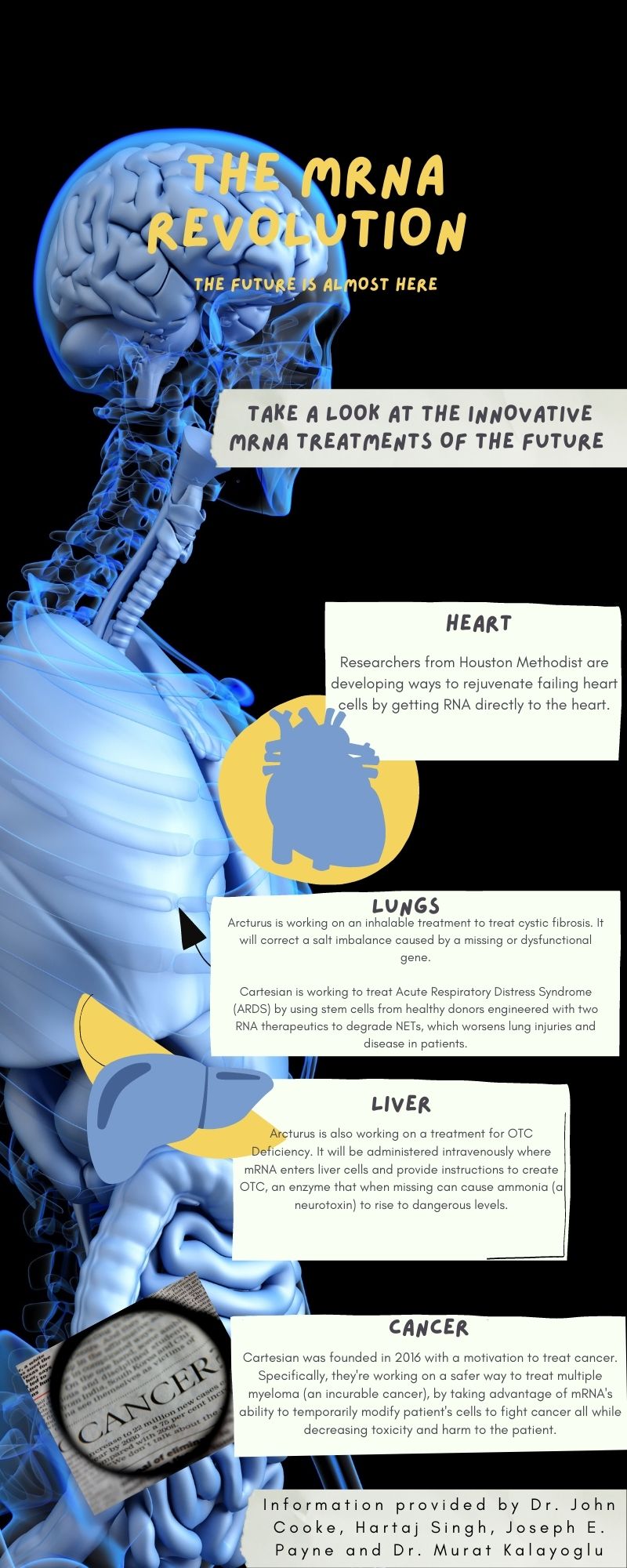
Because it’s easily modified, there are many ways to get RNA into the body besides a shot in the arm. For example, researchers at Houston Methodist are developing a way to rejuvenate failing heart cells by delivering RNA through a catheter or tube directly to the pericardium, the cellophane-like membrane that surrounds the heart.
RNA also has the capacity to reprogram the cells and encourage blood vessel growth, which can enhance the healing properties of collagen scaffolds or a 3D porous network of proteins that acts as a “scaffold” when inserted into wounds. This allows healthy cells to regenerate damaged tissues, says Cooke, like skin and ligaments and tendons — some of the tissues where collagen is found.
By putting mRNA into these structures, they can be used on more tissue types and turn cells that would otherwise scar due to damage from a heart attack, a burn or a severe cut into ones that would enhance blood vessel growth without leaving any scar, according to Cooke.
“The possibilities are endless for applications to any organ system,” he says.
However, even though mRNA is an “amazing drug,” it is also easily degraded by air, light, blood, and saliva, says Joseph E. Payne, chief executive of Arcturus Therapeutics, a California-based maker of mRNA medicines. As a result, he says, “getting it safely and effectively to where it needs to be is quite a challenge.”
Contrary to popular belief, mRNA therapies didn’t come out of nowhere during the pandemic. Depending on who you ask, they have been in development for about 40 years, according to biotech business analyst Hartaj Singh, a managing director at Oppenheimer & Co. in New York.
What finally made it a true contender for creating novel treatments was work done about 17 years ago by researchers Dr. Drew Weissman and Katalin Karikó — work that led directly to the development of the Pfizer/BioNTech and Moderna vaccines.
The University of Pennsylvania-based researchers cleared a hurdle that had plagued other RNA-therapy hopefuls for over 20 years: They figured out how to modify the genetic coding in mRNA so that it would not be attacked by the immune system when introduced into the body.
Thanks to that modification, biotech companies like Arcturus and Maryland-based Cartesian Therapeutics have been able to create mRNA therapies that don’t trigger inflammation and other harmful immune responses. Researchers at Cartesian are combining RNA and cell therapy to use the cell not only as a factory to produce RNA, but also as a vehicle to deliver the therapeutics directly to the affected site to treat respiratory diseases, autoimmune disorders and even cancer, said Cartesian’s chief executive officer, Dr. Murat Kalayoglu. Using living cells to deliver RNA differs from the approach taken by companies like Moderna and Pfizer/BioNTech for their COVID-19 vaccines, Kalayoglu explains, which use tiny fat-based or lipid nanoparticles to package the altered RNA and deliver it into the body.
[Credit: Deborah Balthazar]
BioNTech, which was founded in 2008 to develop individualized cancer therapies, also uses lipid nanoparticles to deliver its cancer vaccines and personalized immunotherapies; four of which are currently in clinical trials. The company’s FixVac or “fixed vaccine” platform lists a potential therapy they developed for metastatic melanoma: Out of 22 people given the experimental treatment, there have been four partial responses to the vaccine. The disease has remained stable in eight additional cases. Other vaccine candidates currently in clinical trials under FixVac include drugs for head and neck cancer, as well as early-stage prostate and ovarian cancers.
Companies like Cartesian are also trying to take advantage of another quirk of mRNA: It doesn’t last long in the body, so the cell modifications are temporary. “There’s a self-limiting ability to express the protein when you engineer a cell with RNA. And then, because of that, you can make your treatment much safer,” especially for early-stage cancers, according to Cartesian’s Kalayoglu. While permanently modifying a cell would be useful for advanced cancers, it would also increase the toxicity of the treatment and increase the risk of harm to the patient.
Cartesian is also working on using mRNA to treat myasthenia gravis, an autoimmune disorder that weakens skeletal muscles, resulting in drooping eyelids or slurred speech. Researchers are using mRNA to modify patients’ own infection-fighting T-cells, redirecting them to kill the cells that are producing the antibodies that attack the patient’s own body and cause muscle weakness.
Myasthenia gravis is rare, with approximately 36,000 to 60,000 cases in the U.S. But it’s not unusual for a young company like Cartesian to target a rare disease: There’s less competition with other companies and it’s easier to acquire the necessary ingredients in smaller quantities, according to Singh, the biotech analyst.
Similarly, Arcturus is tackling rare diseases like Ornithine Transcarbamylase (OTC) Deficiency which impacts the liver and affects over 10,000 people worldwide. OTC is an enzyme that when missing can cause ammonia, a neurotoxin, to rise to dangerous levels.
“Ammonia can cross the blood-brain barrier and cause significant damage to your brain,” Payne says, adding that the neurotoxin is found in products like Windex. “You can imagine that’s not good.”
To get the therapeutic into the liver, the treatment is administered intravenously. Protected by fat, the mRNA travels through the bloodstream and sticks to a protein found on the surface of hepatocytes or specific liver cells. The mRNA enters the cells with the protein and from there will provide instructions to the cells to create OTC.
Arcturus is also working on treating cystic fibrosis, which though classified as a rare disease, is more well known and affects over 30,000 Americans and between 85,000 and 100,000 people worldwide. This treatment, which has been shown to be effective in ferrets, primates and mice, according to Payne, is inhaled through the lungs to correct a salt imbalance caused by a missing or dysfunctional gene. The gene-induced salt imbalance leads to the symptoms of cystic fibrosis, including inflammation and difficulty breathing.
Companies like Moderna and BioNTech have a big advantage because their successful vaccine work has allowed them to build expertise as well as credibility with investors. They now have the capital they need to build production facilities and scale up their supply chain, says Singh.
Payne says investors will continue to be interested in mRNA companies because there are currently so few companies in the space. So will larger drug manufacturers like Sanofi, which recently acquired Translate Bio to accelerate its progress in mRNA vaccines and therapeutics.
Singh doesn’t expect many of the mRNA drugs being developed by small firms to reach the marketplace for another three to five years. Even so, he says, they’re coming.
“I fully expect that there will be many, many messenger RNA companies in the future. But we’re going to benefit from being one of the first and so that’s what’s exciting,” says Payne.
Cartesian’s 22 researchers, meanwhile, are working on relatively rare conditions such as myasthenia gravis, Acute Respiratory Distress Syndrome and multiple myeloma. But while early trials have generated positive results, Kalayoglu recognizes that the company has a long way to go. Despite the exciting possibilities of mRNA therapy, it’s important to work slowly and deliberately to make sure that the technology will benefit patients, he adds.
“I’m humbled by how much work we need to do,” says Kalayoglu, “and how hard we need to work in order to get to where the most number of patients can benefit from this.”
1 Comment
With my amyotrophic lateral sclerosis (ALS), the first thing that happened almost 2 years ago now, was speaking as if I were drunk. I wasn’t. I initially did improve speech (articulating clearly but slow) but now I can no longer speak in an acceptable way. Then, a year later eating became problematic, I was biting my tongue and lips, and chewing became weak and less controlled. Soon after that some fingers started to fail me and things would drop out of my hands. Somewhere at that time bulbar ALS was diagnosed. The Rilutek (riluzole) did very little to help me. The medical team did even less. My decline was rapid and devastating.. We tried every shot available but nothing was working. There has been little if any progress in finding a reliable treatment, Our care provider introduced us to Kycuyu Health Clinic ALS/MND herbal treatment. The treatment is a miracle.i recovered significantly!